
US: (312) 803-0262
Temperature Monitoring Solutions
The Essential Regulatory Platform
JRI MySirius combines real-time temperature monitoring, in-house ISO 17025 calibration, and GxP thermal mapping in one platform — so your facility is always audit-ready.
Continuous temperature, humidity & environmental monitoring
In-house calibration — no external lab, no waiting weeks
Temperature mapping built into the same platform
Cloud or on-premise — your choice
FDA 21 CFR Part 11 · GxP · GMP · FSMA compliant
24/7 alerts: SMS, email & voice call
When an FDA Inspector Walks In, Credentials Matter.
Validated by COFRAC, ILAC MRA, and NIST
Deliver trust that SaaS-only monitoring platforms simply cannot claim.
180+ years of metrology heritage — Founded in Paris in 1845. Precision instrumentation is in our DNA, not bolted on as a feature.
COFRAC / ILAC MRA recognized lab — International accreditation recognized in 100+ countries. Not self-certified.
ANAB-accredited US lab (TMI-USA) — American-accredited local support. Your FDA auditor knows these credentials.
ISO 9001 • ISO 17025 • ISO 27001 • GAMP 5 • 21 CFR Part 11 — Every certification your regulated facility requires.

TRUSTED BY:





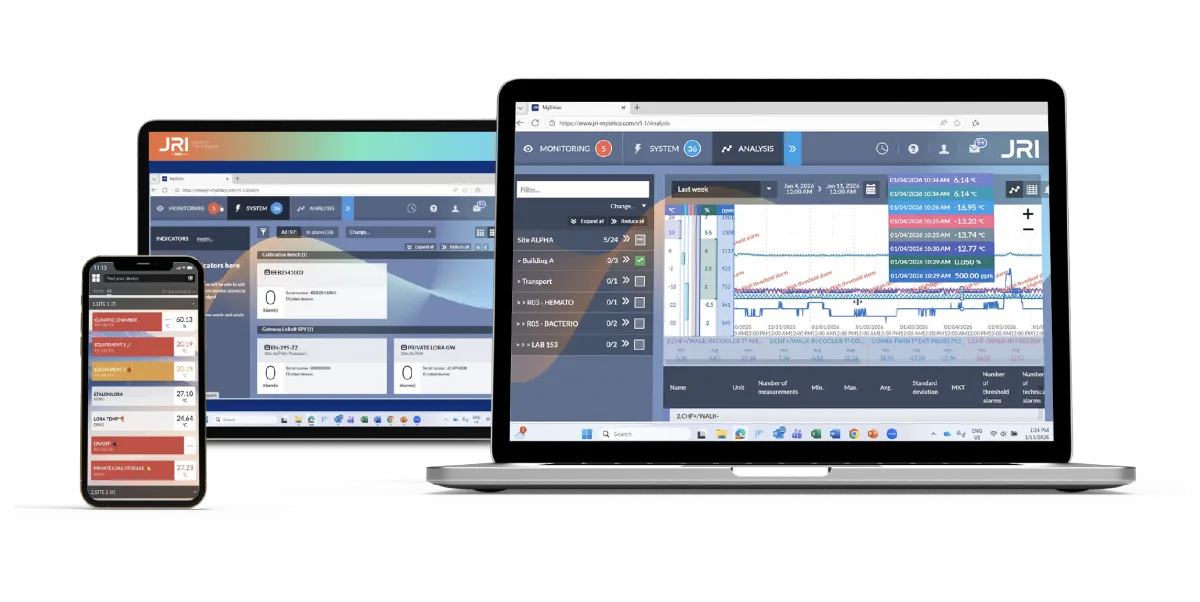
Stop Paying Three Vendors to Do What One Platform Should.
Most temperature monitoring systems stop at the alert. JRI MySirius eliminates the need for a separate calibration service and a separate mapping contractor.
Real-time temperature monitoring — Every sensor, every location, every parameter. Continuous. Never manual.
In-house ISO 17025 calibration — Run calibration yourself inside MySirius. Cut external lab costs permanently.
Thermal mapping built in — Qualify your chambers and cold rooms without a contractor. Your team runs it. Your platform documents it.
One audit trail — Monitoring data, calibration records, and mapping reports all in one tamper-proof system.
MySirius from JRI is built for facilities that cannot afford to fail.
FDA 21 CFR Part 11
COFRAC Certified
GAMP 5 Validated
ISO/IEC 17025 Accredited
Azure Secure Cloud
Temperature Mapping That Doesn't Require a Contractor.
JRI's built-in mapping module lets your team perform full GxP-compliant temperature distribution studies — chambers, cold rooms, warehouses — without hiring outside validation specialists.
Meets Every Standard Your Auditor Will Ask About
Ready to See What Complete Temperature Monitoring Compliance Looks Like?
Get a free quote tailored to your facility size, industry, and compliance requirements.
Pharma · Life Sciences · Hospitals · Blood Banks · Research Labs · Food Safety
This site is not affiliated with Facebook, Google, or any social media platform. JRI is a subsidiary of MMS Group. MySirius is a registered platform of Jules Richard Instruments.